Dental Implant Motor Brushless Surgical Machine Drill Motor with 201 Handpiece


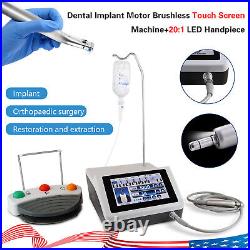








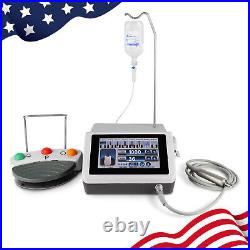











Torque range: 5-70Ncm(gear ratio:20:1). Size: diameter 24mm, long 110mm. We commited ourselves to your 100% satisfaction both with our goods and services. Feedback if you are satisfied with our items. Me immediately before leave us neutral feedback or negtive feedback, We will try our best to resolve any problem for you untill you are satisfied. Pls let me know before taking any action, because your action will has an important impact on our shop. So pls give me a chance to offer the best sollution for you!
Company name: Dreamland Technology Ltd. Return within 30 days is accepted, though the item in good condition. Food & Drug Administration and state and local regulatory agencies.
If you have questions about legal obligations regarding sales of medical devices, you should consult with the FDA's Center for Devices and Radiological Health. This dental items is not meant to replace your doctor diagnostic. It is mainly for your piece of mind and comfort. Regulation Number: 21 CFR 872.6070. FDA for Handpiece : 510(K) Number: K181691, Regulation Number: 872.4200, Classification Product Code: EFB, Subsequent Product Code: EGS.FDA for Scaler : 510(K) Number: K163414, Regulation Number: 872.4850, Product Code: ELC.